Empower Your Clinical Research With Us
Unlock Your Potential By Joining Our Clinical Research Network!
Accelerating Evidence-Based Health Through Collaborative Clinical Research
At the Clinical Research Network Centre (CLIREN Centre), we accelerate high-impact health research by connecting investigators, institutions, and communities through a robust, integrated research infrastructure. We champion collaboration, data-driven methodologies, and ethical clinical practices to generate timely, relevant, and high-quality evidence that informs policy, improves patient care, and strengthens health systems.
By leveraging our expansive network of partners and a commitment to capacity building, we serve as a hub for excellence in multi-site clinical trials, health innovation, and knowledge translation. Our strategic focus is to support equitable and sustainable research ecosystems that address priority health challenges locally and globally.
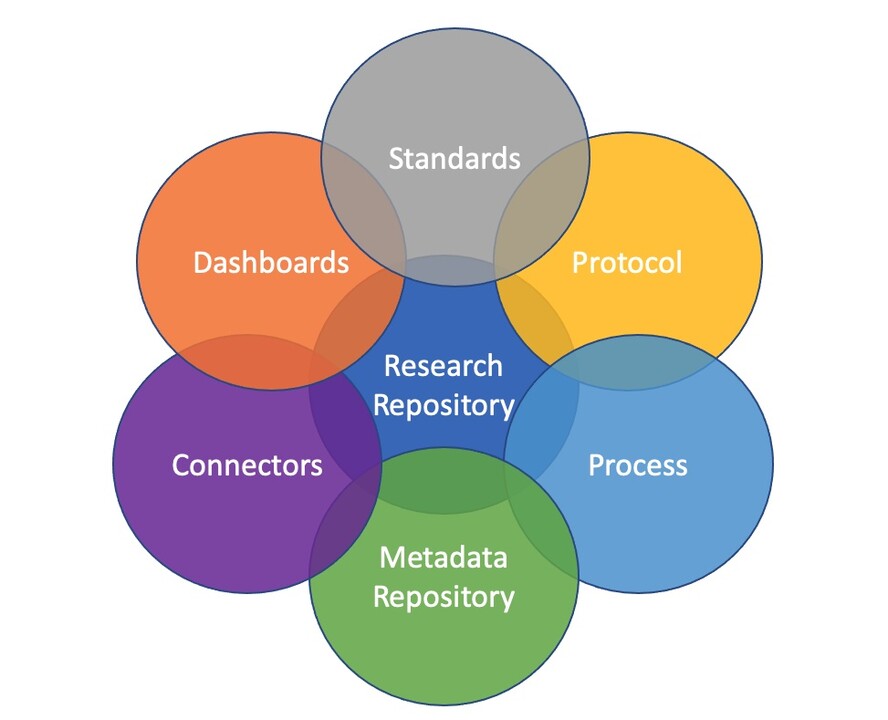
Our Research Platform
At CLIREN Centre, we provide the organizational and technical infrastructure to support multi-site clinical studies, including:
- Data management systems (e.g., REDCap, ODK, & other EDC platforms)
-
Statistical Support
-
Reports Generation and Data Dashboards
-
Clinical Research Monitoring
- Regulatory Support
- Project Management
- Laboratory Support
- Grant Management & Support
Our Capacity Building
To sustain research excellence, we provide the following training services: –
-
Data Management for Clinical Research – Comprehensive training on data collection methods, database design, data quality assurance, cleaning techniques, and data security — tailored for clinical research environments.
-
Practical Clinical Research Training – Short courses and workshops on Good Clinical Practice (GCP), protocol adherence, participant safety, data integrity, and emerging research methodologies.
-
Descriptive & Inferential Statistics – Practical, hands-on workshops teaching researchers how to manage, clean, analyze, and visualize clinical trial and observational study data using R programming. Topics include working with EDC exports, data wrangling (using
dplyr,tidyr), producing clean analysis datasets, and basic reporting with R Markdown and R Shiny. - Good Clinical & Laboratory Practice (GCLP) – Tailored programs to assess laboratory capabilities, support accreditation processes (e.g., ISO 15189), and build sustainable quality systems.

We help organizations GAIN more through…

Our Research Platform
At CLiREN Centre, we offer a comprehensive platform that supports every stage of the clinical research journey. Our services include:
Our Portfolio
Centres of Excellence
Mentorships Done
Training Cohorts
%
Project Completion Rate
Blogs
Contact
+254 782 555 006
The Clinical Research Network,
Bishop Magua Building
5th Floor.
Off Ngong Road, Nairobi
